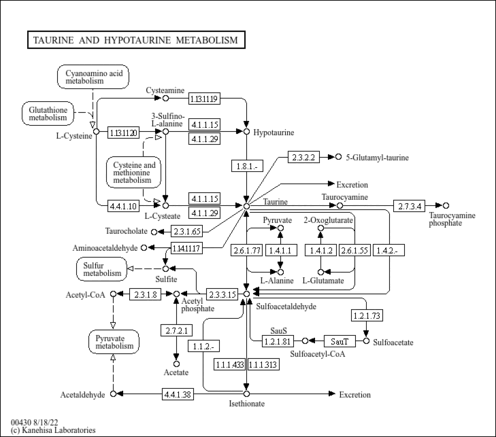
| General Reference | - Johnston, M., Hillier, L., Riles, L., Albermann, K., Andre, B., Ansorge, W., Benes, V., Bruckner, M., Delius, H., Dubois, E., Dusterhoft, A., Entian, K. D., Floeth, M., Goffeau, A., Hebling, U., Heumann, K., Heuss-Neitzel, D., Hilbert, H., Hilger, F., Kleine, K., Kotter, P., Louis, E. J., Messenguy, F., Mewes, H. W., Hoheisel, J. D., et, a. l. .. (1997). "The nucleotide sequence of Saccharomyces cerevisiae chromosome XII." Nature 387:87-90.9169871
- Penninckx, M., Jaspers, C., Wiame, J. M. (1980). "Glutathione metabolism in relation to the amino-acid permeation systems of the yeast Saccharomyces cerevisiae. Occurrence of gamma-glutamyltranspeptidase: its regulation and the effects of permeation mutations on the enzyme cellular level." Eur J Biochem 104:119-123.6102906
- Payne, G. M., Payne, J. W. (1984). "gamma-Glutamyltransferase is not involved in the bulk uptake of amino acids, peptides or gamma-glutamyl-amino acids in yeast (Saccharomyces cerevisiae)." Biochem J 218:147-155.6143552
- Jaspers, C. J., Penninckx, M. J. (1984). "Glutathione metabolism in yeast Saccharomyces cerevisiae. Evidence that gamma-glutamyltranspeptidase is a vacuolar enzyme." Biochimie 66:71-74.6143574
- Elskens, M. T., Jaspers, C. J., Penninckx, M. J. (1991). "Glutathione as an endogenous sulphur source in the yeast Saccharomyces cerevisiae." J Gen Microbiol 137:637-644.1674526
- Lussier, M., White, A. M., Sheraton, J., di Paolo, T., Treadwell, J., Southard, S. B., Horenstein, C. I., Chen-Weiner, J., Ram, A. F., Kapteyn, J. C., Roemer, T. W., Vo, D. H., Bondoc, D. C., Hall, J., Zhong, W. W., Sdicu, A. M., Davies, J., Klis, F. M., Robbins, P. W., Bussey, H. (1997). "Large scale identification of genes involved in cell surface biosynthesis and architecture in Saccharomyces cerevisiae." Genetics 147:435-450.9335584
- Mehdi, K., Penninckx, M. J. (1997). "An important role for glutathione and gamma-glutamyltranspeptidase in the supply of growth requirements during nitrogen starvation of the yeast Saccharomyces cerevisiae." Microbiology 143 ( Pt 6):1885-1889.9202464
- Mehdi, K., Thierie, J., Penninckx, M. J. (2001). "gamma-Glutamyl transpeptidase in the yeast Saccharomyces cerevisiae and its role in the vacuolar transport and metabolism of glutathione." Biochem J 359:631-637.11672438
- Springael, J. Y., Penninckx, M. J. (2003). "Nitrogen-source regulation of yeast gamma-glutamyl transpeptidase synthesis involves the regulatory network including the GATA zinc-finger factors Gln3, Nil1/Gat1 and Gzf3." Biochem J 371:589-595.12529169
- Ghaemmaghami, S., Huh, W. K., Bower, K., Howson, R. W., Belle, A., Dephoure, N., O'Shea, E. K., Weissman, J. S. (2003). "Global analysis of protein expression in yeast." Nature 425:737-741.14562106
- Kumar, C., Sharma, R., Bachhawat, A. K. (2003). "Investigations into the polymorphisms at the ECM38 locus of two widely used Saccharomyces cerevisiae S288C strains, YPH499 and BY4742." Yeast 20:857-863.12868055
|
|---|