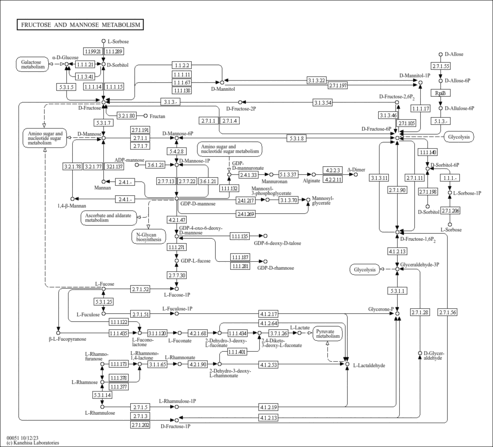
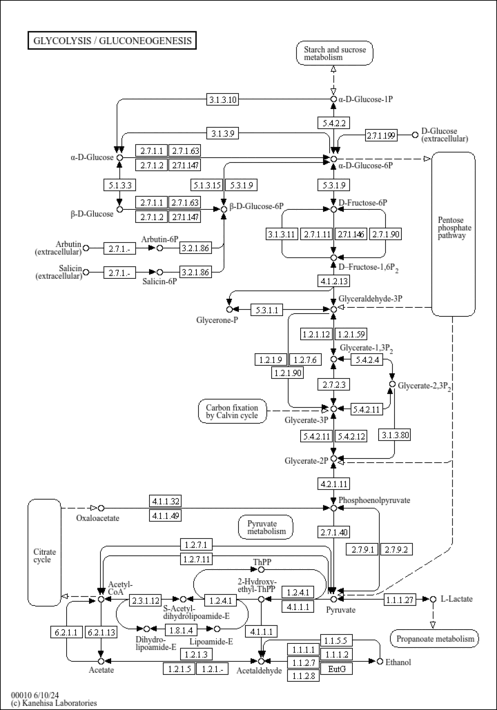
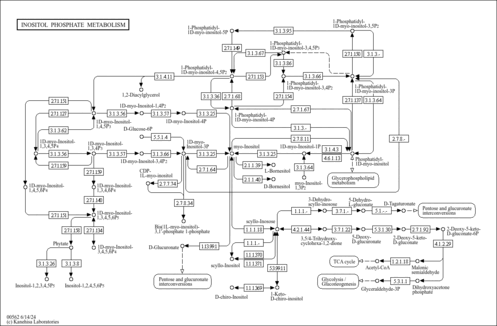
| General Reference | - Alber, T., Kawasaki, G. (1982). "Nucleotide sequence of the triose phosphate isomerase gene of Saccharomyces cerevisiae." J Mol Appl Genet 1:419-434.6759603
- Jacq, C., Alt-Morbe, J., Andre, B., Arnold, W., Bahr, A., Ballesta, J. P., Bargues, M., Baron, L., Becker, A., Biteau, N., Blocker, H., Blugeon, C., Boskovic, J., Brandt, P., Bruckner, M., Buitrago, M. J., Coster, F., Delaveau, T., del Rey, F., Dujon, B., Eide, L. G., Garcia-Cantalejo, J. M., Goffeau, A., Gomez-Peris, A., Zaccaria, P., et, a. l. .. (1997). "The nucleotide sequence of Saccharomyces cerevisiae chromosome IV." Nature 387:75-78.9169867
- Hu, Y., Rolfs, A., Bhullar, B., Murthy, T. V., Zhu, C., Berger, M. F., Camargo, A. A., Kelley, F., McCarron, S., Jepson, D., Richardson, A., Raphael, J., Moreira, D., Taycher, E., Zuo, D., Mohr, S., Kane, M. F., Williamson, J., Simpson, A., Bulyk, M. L., Harlow, E., Marsischky, G., Kolodner, R. D., LaBaer, J. (2007). "Approaching a complete repository of sequence-verified protein-encoding clones for Saccharomyces cerevisiae." Genome Res 17:536-543.17322287
- Alber, T., Hartman, F. C., Johnson, R. M., Petsko, G. A., Tsernoglou, D. (1981). "Crystallization of yeast triose phosphate isomerase from polyethylene glycol. Protein crystal formation following phase separation." J Biol Chem 256:1356-1361.7005233
- Garrels, J. I., Futcher, B., Kobayashi, R., Latter, G. I., Schwender, B., Volpe, T., Warner, J. R., McLaughlin, C. S. (1994). "Protein identifications for a Saccharomyces cerevisiae protein database." Electrophoresis 15:1466-1486.7895733
- Ghaemmaghami, S., Huh, W. K., Bower, K., Howson, R. W., Belle, A., Dephoure, N., O'Shea, E. K., Weissman, J. S. (2003). "Global analysis of protein expression in yeast." Nature 425:737-741.14562106
- Chi, A., Huttenhower, C., Geer, L. Y., Coon, J. J., Syka, J. E., Bai, D. L., Shabanowitz, J., Burke, D. J., Troyanskaya, O. G., Hunt, D. F. (2007). "Analysis of phosphorylation sites on proteins from Saccharomyces cerevisiae by electron transfer dissociation (ETD) mass spectrometry." Proc Natl Acad Sci U S A 104:2193-2198.17287358
- Albuquerque, C. P., Smolka, M. B., Payne, S. H., Bafna, V., Eng, J., Zhou, H. (2008). "A multidimensional chromatography technology for in-depth phosphoproteome analysis." Mol Cell Proteomics 7:1389-1396.18407956
- Lolis, E., Alber, T., Davenport, R. C., Rose, D., Hartman, F. C., Petsko, G. A. (1990). "Structure of yeast triosephosphate isomerase at 1.9-A resolution." Biochemistry 29:6609-6618.2204417
- Lolis, E., Petsko, G. A. (1990). "Crystallographic analysis of the complex between triosephosphate isomerase and 2-phosphoglycolate at 2.5-A resolution: implications for catalysis." Biochemistry 29:6619-6625.2204418
- Wierenga, R. K., Noble, M. E., Davenport, R. C. (1992). "Comparison of the refined crystal structures of liganded and unliganded chicken, yeast and trypanosomal triosephosphate isomerase." J Mol Biol 224:1115-1126.1569570
- Alber, T. C., Davenport, R. C. Jr, Giammona, D. A., Lolis, E., Petsko, G. A., Ringe, D. (1987). "Crystallography and site-directed mutagenesis of yeast triosephosphate isomerase: what can we learn about catalysis from a "simple" enzyme?" Cold Spring Harb Symp Quant Biol 52:603-613.3331346
- Jogl, G., Rozovsky, S., McDermott, A. E., Tong, L. (2003). "Optimal alignment for enzymatic proton transfer: structure of the Michaelis complex of triosephosphate isomerase at 1.2-A resolution." Proc Natl Acad Sci U S A 100:50-55.12509510
|
|---|